Pancreaticoduodenal Artery Aneurysm
A visceral aneurysm built not from wall failure, but from flow — and the decisions that followed had to reflect exactly that.
Clinical Background
A 43-year-old Bahraini male referred to the vascular surgery clinic following incidental identification of a visceral aneurysm on prior cross-sectional imaging. Background of recent ischaemic stroke with right internal carotid artery occlusion, dyslipidaemia, and impaired glucose tolerance. Active smoker — cigarettes and sheesha. On aspirin and statin therapy at the time of referral. No prior abdominal surgery, no history of pancreatitis, and no connective tissue disease documented.
Clinical Presentation
The patient had been experiencing abdominal pain prior to presentation — a symptom that was most probably related to the aneurysm itself. The aneurysm was first identified incidentally on CT performed for a separate indication, but the symptom history added clinical weight to what imaging might otherwise have framed as a routine incidental finding. At the initial vascular clinic review, an urgent CT angiogram was requested to characterise the lesion and interrogate the visceral anatomy in full.
Examination Findings
Haemodynamics: stable
Abdomen: soft, no pulsatile mass
Peripheral pulses: intact bilaterally
Peritonism: none
Investigations
Pre-Procedural Bloods — Arranged
| Investigation | Rationale |
|---|---|
| Full Blood Count | Baseline haemoglobin; exclude occult anaemia |
| Renal Profile | Contrast clearance assessment pre-procedure |
| Coagulation Profile | Pre-interventional status; patient on antiplatelet therapy |
| ECG + Chest X-Ray | Cardiorespiratory pre-procedural assessment |
CT Angiography — Abdomen, Pelvis and Aorta
The CTA confirmed the clinical suspicion and mapped the haemodynamic architecture behind it. The aorta was of normal calibre with no significant atherosclerotic burden. The coeliac artery demonstrated severe stenosis at its origin with a characteristic hook morphology on sagittal reconstruction — the hallmark of extrinsic compression by the median arcuate ligament. The main SMA stem was patent throughout.
Arising from the inferior pancreaticoduodenal artery was a saccular aneurysm measuring 19 × 16 × 14 mm with a narrow 3 mm neck. Partial wall calcification and partial luminal thrombosis were present — features that together speak to a lesion that is neither new nor static. Jejunal, ileal, and right ileocolic branches were patent. Renal arteries, IMA, and the iliofemoral axes were all unremarkable. No intraperitoneal or retroperitoneal haematoma was identified.
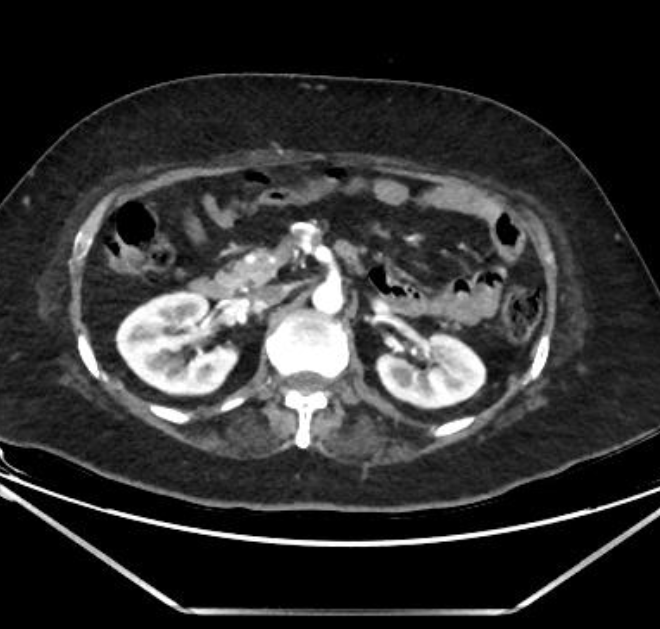
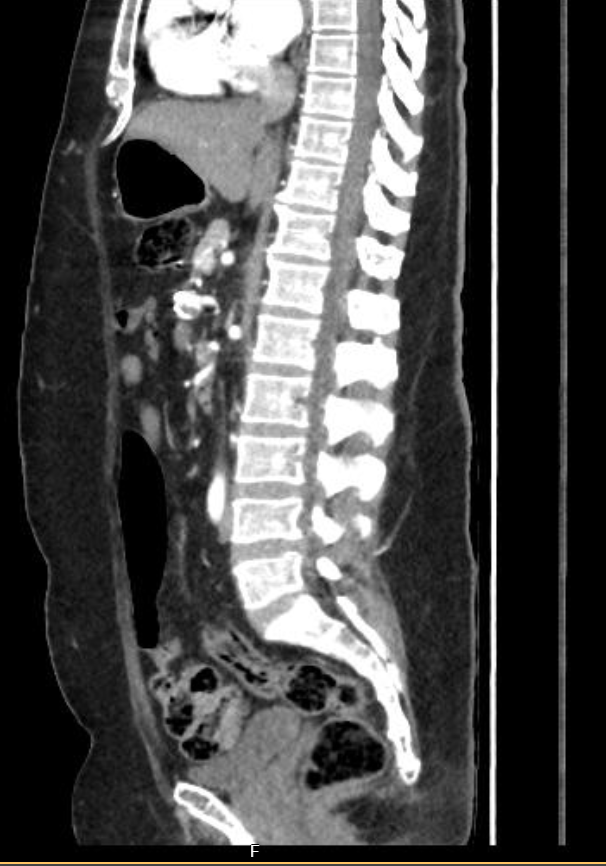
CTA — Inferior pancreaticoduodenal artery saccular aneurysm with coeliac origin stenosis
Vascular Interpretation
This is a flow-related inferior pancreaticoduodenal artery aneurysm. Severe proximal coeliac stenosis — consistent with median arcuate ligament compression given the hook morphology and the absence of significant aortic atherosclerosis in a young patient — has redirected perfusion pressure through the SMA–pancreaticoduodenal arcade. The chronic hyperdynamic collateral flow is the engine behind aneurysm formation. The saccular morphology, mural calcification, and partial thrombosis confirm that this is not a benign incidental finding: it is a haemodynamic lesion with meaningful rupture risk.
Clinical Impression
The diagnosis here is not simply an aneurysm — it is the downstream signature of an upstream problem. Severe coeliac origin stenosis, almost certainly from median arcuate ligament compression, has forced the SMA–pancreaticoduodenal arcade to carry a haemodynamic burden it was not built to sustain. The inferior pancreaticoduodenal artery has compensated — and in doing so, has dilated into a saccular aneurysm that now carries its own independent risk.
The turning point in the clinical reasoning was recognising the hook morphology of the coeliac stenosis. In a 43-year-old with no significant aortoiliac atherosclerosis, this pattern points strongly to extrinsic rather than intrinsic disease — and that distinction is not an incidental anatomical observation. It determines the treatment sequence, the technique, and the risk of getting the order wrong. Treat the aneurysm in isolation and you leave the coeliac problem in place. Stent the coeliac without releasing the ligament and the stent will eventually fracture under ongoing extrinsic compression. The two problems share a single haemodynamic story, and the management plan had to reflect that.
Primary Diagnosis
Inferior pancreaticoduodenal artery saccular aneurysm (19 × 16 × 14 mm), with partial mural calcification and partial luminal thrombosis, in the setting of severe coeliac origin stenosis consistent with median arcuate ligament compression.
Management Plan
Phase 1 — Assessment and MDT Planning
Following urgent CTA, the case was reviewed in the vascular surgery clinic and brought to a formal multidisciplinary team discussion with interventional radiology. Pre-procedural workup was arranged: FBC, renal profile, coagulation profile, ECG, and chest X-ray. Anaesthetic assessment was requested. Vascular risk factor optimisation was initiated alongside — smoking cessation, lipid and glucose control, continuation of antiplatelet therapy. Red flag symptoms were clearly documented and explained to the patient.
Phase 2 — Endovascular Coiling (Completed)
The MDT decision — reached jointly with the interventional radiology team — was to proceed with endovascular coil embolisation of the aneurysm as the first priority. The rationale was clear: the life-threatening risk was the aneurysm. With a narrow 3 mm neck, the anatomy was technically suitable for superselective coiling using the isolation technique, packing proximal and distal to the sac. During the procedure, superselective DSA was performed to confirm that the isolation technique successfully excluded the aneurysm sac while maintaining robust flow through the adjacent pancreaticoduodenal collaterals, thereby ensuring no compromise to hepatic or splenic inflow. The procedure was carried out successfully in collaboration with the IR team, with confirmed aneurysm exclusion.
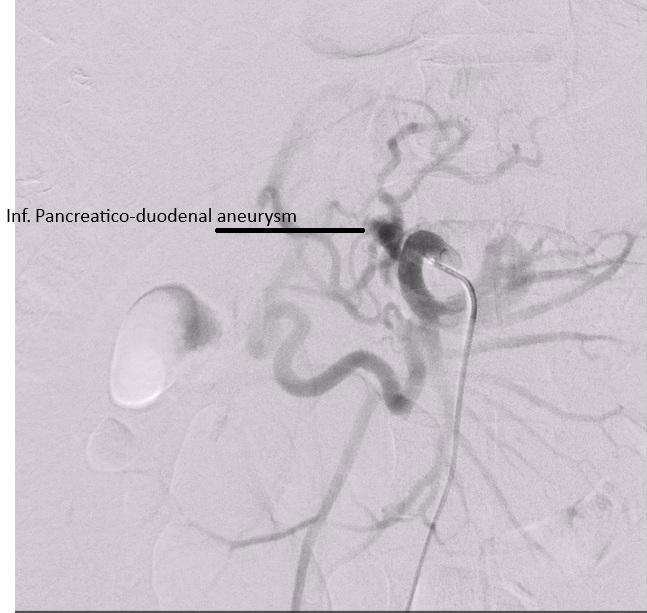
DSA — Inferior pancreaticoduodenal artery aneurysm prior to embolisation
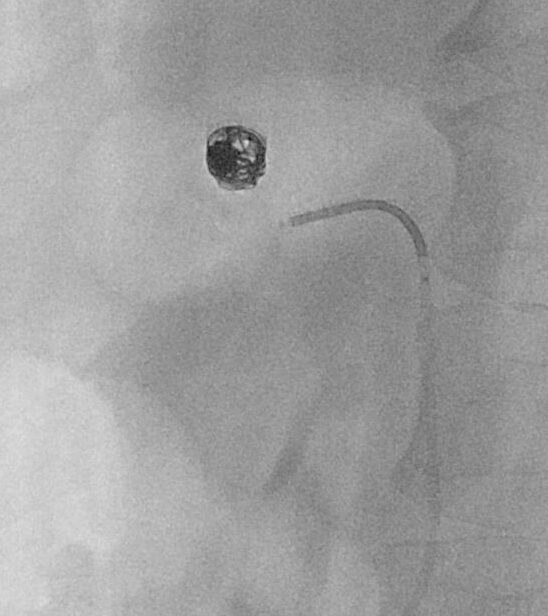
DSA — Post-embolisation: coil pack in situ, aneurysm excluded
Phase 3 — Staged Laparoscopic MAL Release (Planned)
With the immediate life-threatening risk resolved, the underlying cause — median arcuate ligament compression of the coeliac axis — remains to be addressed. Following a further MDT discussion, the plan is for laparoscopic median arcuate ligament dissection as a staged procedure. Addressing the extrinsic coeliac compression reduces the long-term haemodynamic burden on the SMA–pancreaticoduodenal arcade and mitigates the risk of recurrent aneurysm formation or aneurysm reperfusion. This sequence — fix the life-threatening problem first, address the root cause second — reflects sound surgical prioritisation.
Phase 4 — Surveillance
Post-embolisation surveillance imaging per joint vascular and IR plan, assessing for aneurysm reperfusion and vascular remodelling at 1–2 year intervals in line with SVS recommendations. Following MAL release, reassessment of coeliac patency and haemodynamic normalisation of the pancreaticoduodenal arcade. Ongoing cardiovascular risk factor management in the context of stroke history and right ICA occlusion.
Teaching Points & Discussion
What does this case teach — and what does the literature say about it?
The Disease
Visceral artery aneurysms are rare — reported in 0.01–0.2% of the general population. Pancreaticoduodenal artery aneurysms account for approximately 2% of all visceral aneurysms, making them a rare but clinically consequential entity. Their importance is disproportionate to their incidence: the risk of rupture approaches 50%, and crucially, that rupture risk is independent of aneurysm size.
This is what makes PDAAs different from most other vascular lesions, where size is the dominant trigger for intervention. Here, a 15 mm aneurysm carries the same imperative to treat as a 25 mm one. The reason is aetiology: most PDAAs are not degenerative. They are haemodynamic — built from flow, not from wall failure. A vessel under chronic hyperdynamic stress is unpredictable in a way that a slowly expanding atherosclerotic aneurysm is not.
The mechanism in the majority of cases is coeliac axis obstruction — whether from intrinsic atherosclerosis or extrinsic compression by the median arcuate ligament. The SMA–pancreaticoduodenal arcade becomes the dominant route for hepatic and splenic perfusion. With that comes chronically elevated flow, turbulence, wall stress, and in time, aneurysmal dilatation. A pseudoaneurysm arising in the context of pancreatitis tells a different story: enzymatic digestion of the arterial wall, rupture rates up to 76%, and a patient who is usually already unwell. Distinguishing the two shapes urgency, technique, and the risk calculus of intervention itself.
What the Literature Says
The evidence base for PDAAs is built from case series, systematic reviews, and registry data — there are no randomised trials, and there never will be. The condition is too rare and the rupture stakes too high. What we have is a body of literature that tells a remarkably consistent story about aetiology, intervention thresholds, and the hazards of incomplete anatomical thinking.
SVS Clinical Practice Guidelines — Chaer et al., J Vasc Surg 2020
The Society for Vascular Surgery recommends treatment of all gastroduodenal and pancreaticoduodenal artery aneurysms regardless of size (Grade 1, Quality B) — a deliberate departure from the ≥2 cm threshold applied to most other visceral aneurysms. For both intact and ruptured lesions, coil embolisation is the recommended treatment of choice, with covered stenting or stent-assisted coiling as alternatives where direct embolisation is not anatomically feasible (Grade 2, Quality C). Post-intervention surveillance imaging is recommended every 1–2 years to assess for reperfusion and vascular remodelling.
Coeliac Association — Kubota et al., Ann Gastroenterol Surg 2023
PDAAs are associated with coeliac artery stenosis or occlusion in 50–80% of patients. The coeliac obstruction leads to increased SMA-to-PDA flow, which not only creates the haemodynamic environment for aneurysm formation but also makes the PDA an active and critical collateral supplying hepatic perfusion. This has a direct bearing on intervention strategy: embolise the aneurysm without securing hepatic inflow, and you risk acute ischaemic liver failure. The aneurysm and the coeliac lesion must be considered together — not sequentially, not independently.
MALS and Treatment Failure — Hamlin et al., Ann Vasc Surg 2024
Hamlin’s series illustrates what happens when MALS is not recognised before coeliac intervention. Endovascular stenting of the coeliac artery without prior ligament release carries a high risk of stent fracture — the ligament continues to compress, the stent fatigues, and the coeliac re-occludes. Aneurysm reperfusion follows. The lesson is not that endovascular treatment is contraindicated in MALS; it is that stenting alone, without addressing the extrinsic compression, is not a durable strategy. Ligament release — open or laparoscopic — must precede or accompany definitive coeliac intervention in this subset of patients.
Staged Strategy — Ghoneim et al., Vascular 2024 / Sgroi et al., J Vasc Surg Cases 2023
Both series support a staged approach when the coeliac lesion complicates direct aneurysm embolisation. Where hepatic perfusion is dependent on the pancreaticoduodenal collateral, securing inflow before aneurysm exclusion is the safer sequence. In MALS-related cases specifically, robotic or laparoscopic MAL release followed by coeliac reassessment and then aneurysm treatment achieves both goals without the failure mode of isolated endovascular stenting. This is precisely the pathway now planned for this patient — coiling first to eliminate the life-threatening risk, MAL release as a staged procedure to address the root cause.
Key Lessons
Size does not predict rupture
Unlike most visceral aneurysms, PDAAs carry a size-independent rupture risk approaching 50%. The SVS mandates treatment of all PDAAs regardless of diameter (Grade 1B). A 19 mm flow-related aneurysm in a young patient is not a watch-and-wait case — it is an indication for intervention, and the absence of symptoms does not change that calculus.
Always interrogate the coeliac axis
Up to 80% of PDAAs are associated with coeliac stenosis or occlusion. Before any embolisation, you must establish whether the PDA is functioning as the dominant collateral supplying the hepatic artery. If it is, occluding the aneurysm without alternative hepatic inflow risks acute liver ischaemia. The aneurysm and the coeliac lesion are one haemodynamic problem — treat them as such.
MALS changes the treatment algorithm
When coeliac stenosis is due to median arcuate ligament compression rather than atherosclerosis, endovascular stenting alone has a high failure rate. The ligament continues to exert extrinsic force, the stent fatigues, and the coeliac re-occludes. Differentiating the aetiology — hook morphology on CTA, clinical context, dynamic duplex — is the decision point that determines whether the patient can be managed endovascularly or requires staged surgical ligament release.
Fix the life-threatening problem first
When a patient presents with both a rupturable aneurysm and an underlying anatomical cause, the sequence matters. Coiling the aneurysm first — eliminating the immediate rupture risk — was the right priority here. Laparoscopic MAL release as a planned second stage addresses the root haemodynamic cause without exposing the patient to a combined procedure burden. This is staged surgical thinking: define the hierarchy of risk, act accordingly, and plan the rest.
References
- Chaer RA, Abularrage CJ, Coleman DM, et al. The Society for Vascular Surgery clinical practice guidelines on the management of visceral aneurysms. J Vasc Surg. 2020;72(1S):3S–39S. doi:10.1016/j.jvs.2020.01.039
- Ghoneim B, Nash C, Akmenkalne L, et al. Staged treatment for pancreaticoduodenal artery aneurysm with coeliac artery revascularisation: case report and systematic review. Vascular. 2024. doi:10.1177/17085381221124991
- Hamlin S, Gallo SR, Petrochko J, Wilson J, Sheth S. Pancreaticoduodenal artery aneurysm complicated by median arcuate ligament syndrome. Ann Vasc Surg. 2024;58:213–217. doi:10.1177/15385744231198934
- Kubota K, Shimizu A, Notake T, et al. Treatment strategies for unruptured pancreaticoduodenal artery aneurysms associated with celiac artery occlusion. Ann Gastroenterol Surg. 2023;7(1):182–189. doi:10.1002/ags3.12609
- Corey MR, et al. Presentation and management of true aneurysms of the pancreaticoduodenal arcade with concomitant celiac artery stenosis using the endovascular approach. J Vasc Surg Cases Innov Tech. 2024. doi:10.1016/j.jvscit.2024.00083
- Sgroi MD, Kabutey NK, Krishnam M, Fujitani RM. A minimally invasive approach for management of pancreaticoduodenal artery and gastroduodenal artery aneurysm with celiac artery occlusion. J Vasc Surg Cases Innov Tech. 2023;9(3). doi:10.1016/j.jvscit.2023.00089
- Marone EM, Rinaldi LF, et al. Current debates in the management of visceral artery aneurysms: where the guidelines collide. J Clin Med. 2023;12(9):3267. doi:10.3390/jcm12093267
- Leone N, Migliari M, Baresi GF, Gennai S. Visceral artery aneurysms: a shred of light on rare entities. Vasc Med. 2025. doi:10.1177/14574969251321936
- Yu YN, Xu YS, et al. Ruptured small pancreaticoduodenal artery aneurysm — clinical features similar to pancreatitis: a case report. Medicine. 2023;102(9):e32821. doi:10.1097/MD.0000000000032821
- Inferior pancreaticoduodenal artery pseudoaneurysm causing biliary obstruction. CJEM/PMC. 2024. PMC11166067.